Safe Contrast Agents for Magnetic Resonance Imaging (MRI)
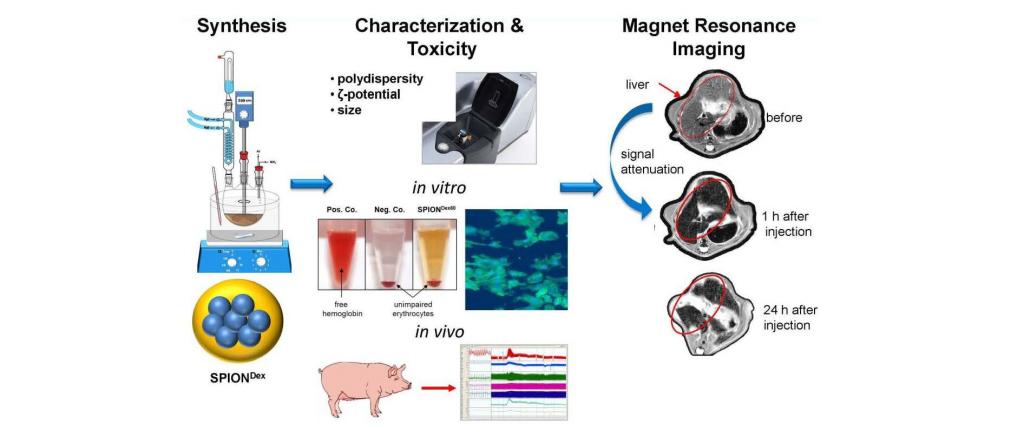
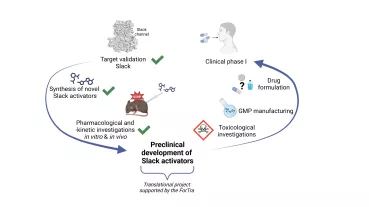
The use of gadolinium-based contrast agents for magnetic resonance imaging (MRI) raises serious safety concerns. Iron oxide nanoparticle-based contrast agents might serve as an alternative, however, currently in Europe no particles are available for this application, and the off-label use of the iron replacement agent ferumoxytol (Feraheme®) as a contrast agent is associated with increased risk to the patients. To address this problem, we developed novel iron oxide nanoparticles (SPIONDex) with dextran coating. Within the present project, we aim to evaluate the safety and the magnetic properties of these particles in comparison with commercially available SPIONs (Feraheme® and Resovist®).
In parallel with the quality-controlled SPIONDex synthesis and upscaling, extensive toxicological and immunological characterization of the tested SPIONs will be performed. The diagnostic accuracy of SPIONDex will be evaluated using in vitro and pre-clinical in vivo MRI in rats. Safety studies in a pig model of hypersensitivity reactions will be performed to facilitate the future approval process with regulatory authorities. The planned investigations are expected to accelerate the clinical translation of the novel MRI contrast agent with improved safety.