
Within the scope of Else Kröner Memorial Fellowships the foundation Else Kröner-Fresenius-Stiftung (EKFS) supports physicians at the beginning of their professional careers who are active in clinical and scientific functions. As a result of being released from clinical responsibilities for a period of two years, these fellowship recipients have the opportunity to focus their efforts on a promising medical research project. Each fellowship is endowed with a total of EUR 250,000.
This year nine medical doctors are receiving an Else Kröner Memorial Fellowship:

Dr. Maximilian Y. Deng, Dept. of Radiation Oncology and Radiation Therapy, Heidelberg University Hospital
Project: Tumor monitoring and molecular risk stratification by means of liquid biopsy in the case of meningeoma patients prior to radiotherapy
Radiotherapy represents an effective treatment approach during therapy for a meningeoma, a brain tumor that arises from the minenges membrane layers. This therapy is particularly preferred in the case of inoperable patients. In the case of hard-to-reach tumors, biopsies are frequently avoided due to the potential risks posed by an operation: the lack of tumor material removes the possibility of a radiation treatment planning adapted to the tumor’s aggressiveness. The project aims to establish so-called exosomes, molecules circulating freely in the bloodstream, as potential biomarkers in the sense of a liquid biopsy for marking the response to therapy and toward the early detection of relapses. The molecular profile of the exosomes will be additionally studied. This is intended to enable a conclusion to be drawn regarding the tumor biology and aggressiveness of the meningeoma. These factors can then be taken into consideration within therapeutic management (e.g. aftercare intervals).

Dr. Jan Kempski, 1st Department of Medicine, University Medical Center Hamburg-Eppendorf (UKE)
Project: Identification of immunological characteristics which influence the response to immunotherapies
The interaction between the immune system and cancer cells has yet to be sufficiently clarified. With the emergence of new immunotherapies, understanding this correlation is becoming more and more important. Immunotherapies can be accompanied by inflammatory reactions which bear similarities to autoimmune diseases and frequently restrict treatment. In this project it is going to be studied how signals from the immune system have an impact on the effectiveness of immunotherapies and which factors lead to a bowel inflammation in the course of these therapies. Especially the interplay of intestinal bacteria and immune cells will be studied. The project objective is to identify characteristics that are able to predict the response to immunotherapies and incidence of inflammatory side effects.

Dr. Dr. Emre Kocakavuk, Dept. of Haemotology and Stem Cell Transplantation, West German Cancer Centre, University Hospital Essen
Project: AI-guided characterization of copy number signatures in the context of cancer therapies
Alongside mutations, copy number variations (CNVs) also have an influence on the onset of cancer, its progression and its resistance to therapy. Dr. Dr. Emre Kocakavuk and his team will be extensively studying CNV signatures in the case of recurring and metastasizing cancerous diseases following treatment. These signatures are going to be interlinked with clinical treatment data. The intention is to use clinical genomic datasets and apply advanced AI- and machine learning algorithms to pave the way for the development of genome-based approaches in personalized oncology.

Dr. Julia Lang-Meli, Department of Medicine II, Medical Center ─ University of Freiburg
Project: Gender-specific disparities in the CD8+ T cell-mediated immune control of chronic hepatitis B viral infection
In the case of chronic hepatitis B viral infections (HBV), it is well known that men exhibit a higher viral load in their blood and more frequently come down with complications such as liver cancer. However, the underlying mechanisms have not been known until now. Experimental data gained from animals and own preliminary work point to a potentially important role played by HBV-specific killer T cells. This research project intends to reveal the immunological mechanisms that lead to a varying clinical progression of chronic HBV infection in men and women. Concretely envisaged for both women and men is a screening in terms of the composition of HBV-specific immune cells along with the definition of the phenotype and function of specific killer T cells, inter alia by means of flow zytometry and scRNA-seq.
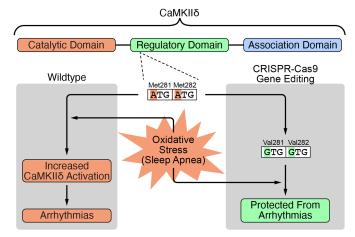
PD Dr. Simon Lebek, Department for Internal Medicine II, University Hospital Regensburg
Project: CRISPR-Cas9 genome editing of CaMKIIδ as antiarrhythmic therapy in the case of sleep-related breathing disorders
Sleep-related breathing disorders are frequently accompanied by cardiac arrythmia and consequently reduce the quality of life and life expectancy, which is why new and improved treatment strategies are urgently needed. PD Dr. Simon Lebek and his team were recently able to show that in the case of patients with sleep-related breathing disorders, an increased activation of a certain disease-associated factor (CaMKIIδ) occurs that leads to cardiac arrythmia. The aim is to deactivate the activation point in this harmful disease-associated factor in order to develop a new treatment strategy. This is intended to be tested and optimized in both human myocardial cells and in live mice with sleep-related breathing disorders. In the long term this approach could lead to a new medication for cardiac arrythmia in patients with sleep-related breathing disorders.

Dr. Caroline Perner, Clinical Dept. for Haematology, Haemostaseology, Oncology and Stem Cell Transplantation, Hannover Medical School (MHH)
Project: Definition of immune cell-dependent and cell-independent physiological changes in sensory neurons during the aging process
Sensory neurons are not only responsible for sensory perceptions, they also actively regulate processes involving the immune system and tissues via the release of neuropeptides and neurotransmitters. They thereby play a decisive role in combating pathogens, in the case of allergic processes, and influence wound healing and regeneration. This project aims at characterizing age-related changes in stimulus recognition and response in sensory neurons and to decipher if immune cells take on an active role during the progression of this process. The findings gained here are intended to help toward a better understanding of the causes for the disrupted equilibrium between tissue and the immune system as aging advances, and to create a starting point for the development of therapies for healthier aging.

Dr. Saskia Räuber, Dept. of Neurology, Düsseldorf University Hospital, Heinrich Heine University Düsseldorf
Project: Analysis of the immune system mechanisms of paraneoplastic neurological syndromes
The immune system constantly attempts to counteract the emergence and spread of cancer cells, and thus the formation and progression of tumors. Nevertheless, this desired immune response can also end up being misrouted and give rise to neurological symptoms. These kinds of disorders termed paraneoplastic neurological syndromes (PNS) can affect different areas of the nervous system. Using modern immunological methods, Dr. Saskia Räuber and her team want to attain a deeper understanding of PNS and fundamental insights regarding immune system behavior in the event of cancerous diseases. In addition to timely diagnosis and therapy for PNS, equally intended as a result is to make a relevant contribution toward the diagnosis and therapy of cancerous diseases at an early stage.
Dr. Marie-Christin Weber, University Hospital “Klinikum rechts der Isar”, Dept. of Surgery, Medical Faculty, Technical University Munich (TUM)
Project: Prevention of intestinal anastomotic insufficiencies via stimulation of serosal wound healing and induction of mesothelial-to-mesenchymal transition of mesothelial cells
Intact intestinal wound healing is the key to a successful surgical therapy in the case of intestinal cancer. Particularly the healing of the suturations that rejoin the two ends of the intestine following tumor resection is decisive. However, the rate of leakages in the region of these suturations (anastomotic insufficiencies) continues to remain high, which leads to high morbidity and mortality. The project being presented is intended to study the cellular processes during anastomotic healing in detail. The emphasis thereby lies on investigating the role of the serosa layer of the intestinal wall and the cells located there, on mesothelial cells, and on anastomotic healing and its potential value for targeted treatments toward the prevention of anastomotic insufficiencies.

Dr. Marcel Seungsu Woo, Institute of Neuroimmunology and Multiple Sclerosis (INIMS), University Medical Center Hamburg-Eppendorf (UKE)
Project: The interferon network in inflammatory neurodegeneration
Multiple sclerosis (MS) is an inflammatory disease of the central nervous system and the most frequent cause of non-traumatic, neurologically based disability during young adulthood. In the case of MS, a chronic inflammation in the brain and spinal cord leads to a continuous loss of nerve cells (neurons) which cannot be treated effectively via therapies. New therapeutic approaches are therefore necessary in order to directly protect the neurons. In this project, various molecular methods are combined to decipher the mechanisms of inflammation-related neurodegeneration, in other words the processes that lead to loss of function in the neurons. Particularly the role that the messenger substance interferon plays in neurodegeneration is intended to be studied and consequently result in the development of new therapeutic approaches.