Combined immunotherapy with peptide vaccination and immune checkpoint inhibition for malignant diseases with DNAJB1-PRKACA gene fusions
Project partner:
Prof. Dr. med. Michael Bitzer, Klinik für Innere Medizin I, Universitätsklinikum Tübingen
Priv.-Doz. Dr. med. Martin Ebinger, Kinderklinik, Universitätsklinikum Tübingen
Prof. Dr. med. Helmut Salih, Klinische Kooperationseinheit Translationale Immunologie, Universitätsklinikum Tübingen
Study Centers:
Universitätsklinikum Freiburg
Universitätsklinikum Ulm
Universitätsklinikum Heidelberg
T cell-based immunotherapy with immune checkpoint inhibitors, CAR-T cells, adoptive T-cell transfer and vaccination strategies are promising thereapeutic options for cancer patients. Neoepitopes of oncogenic gene fusions are a novel superior category of tumor antigens that can be actively targeted by T cell-based immunotherapy.
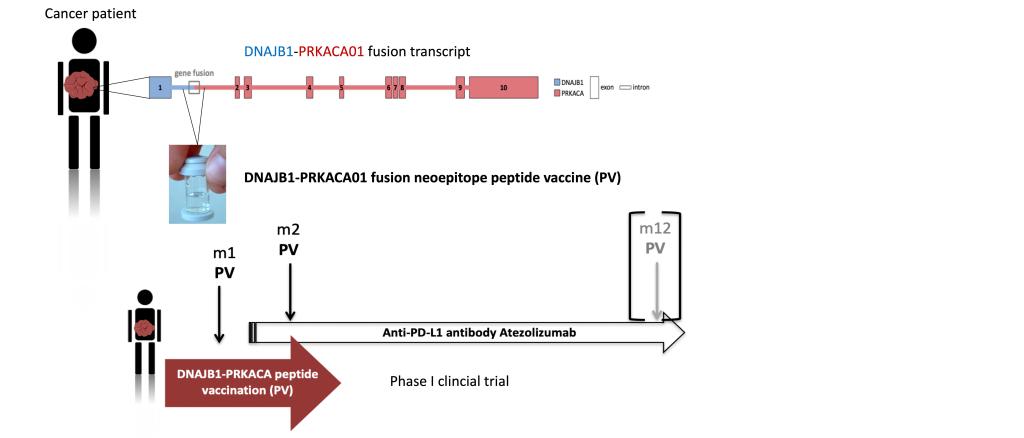
The DNAJB1-PRKACA fusion transcript was identified as the oncogenic driver of tumor pathogenesis in 100% of patients suffering from fibrolamellar hepatocellular carcinoma. Recently, the DNAJB1-PRKACA fusion transcript was also identified in further cancer entities in pancreas and bile duct.
Fusion-VAC-XS15 is a peptide vaccine consisting of a neoepitope of the fusion region DNAJB1-PRKACA adjuvanted with the toll-like receptor (TLR) 1/2 agonist XS15 emulsified in Montanide. Our group recently published data about the immunogenicity ofDNAJB1-PRKACA neoepitopes and first clinical efficacy of a neoepitope-based peptide vaccine (Bauer et al., Nat. commun. 2022).
In the upcoming Phase I basket study financed by ForTra gGmbH the Fusion-VAC-XS15 vaccine will be combined with the anti-PD-L1 immune checkpoint inhibitior atezolizumab to treat patients with advanced cancers carrying the DNAJB1-PRKACA fusion transcript. This projects holds promise to establish gene fusion-targeting immunotherapy as a novel treatment option and to substantially improve the prognosis for cancer patients carrying the DNAJB1-PRKACA fusion transcript.
Here you can find further information.
Details about the study can be found here.




